The Scale of Discovery
From molecular interactions to system-wide network dynamics.
At Neurexpert, we deliver high‑resolution electrophysiology across every level of the nervous system — from isolated molecular targets to intact neuronal circuits. Our integrated platform reveals how compounds behave in real biological systems, providing the mechanistic clarity needed to confidently advance CNS drug discovery. Explore our capabilities below.
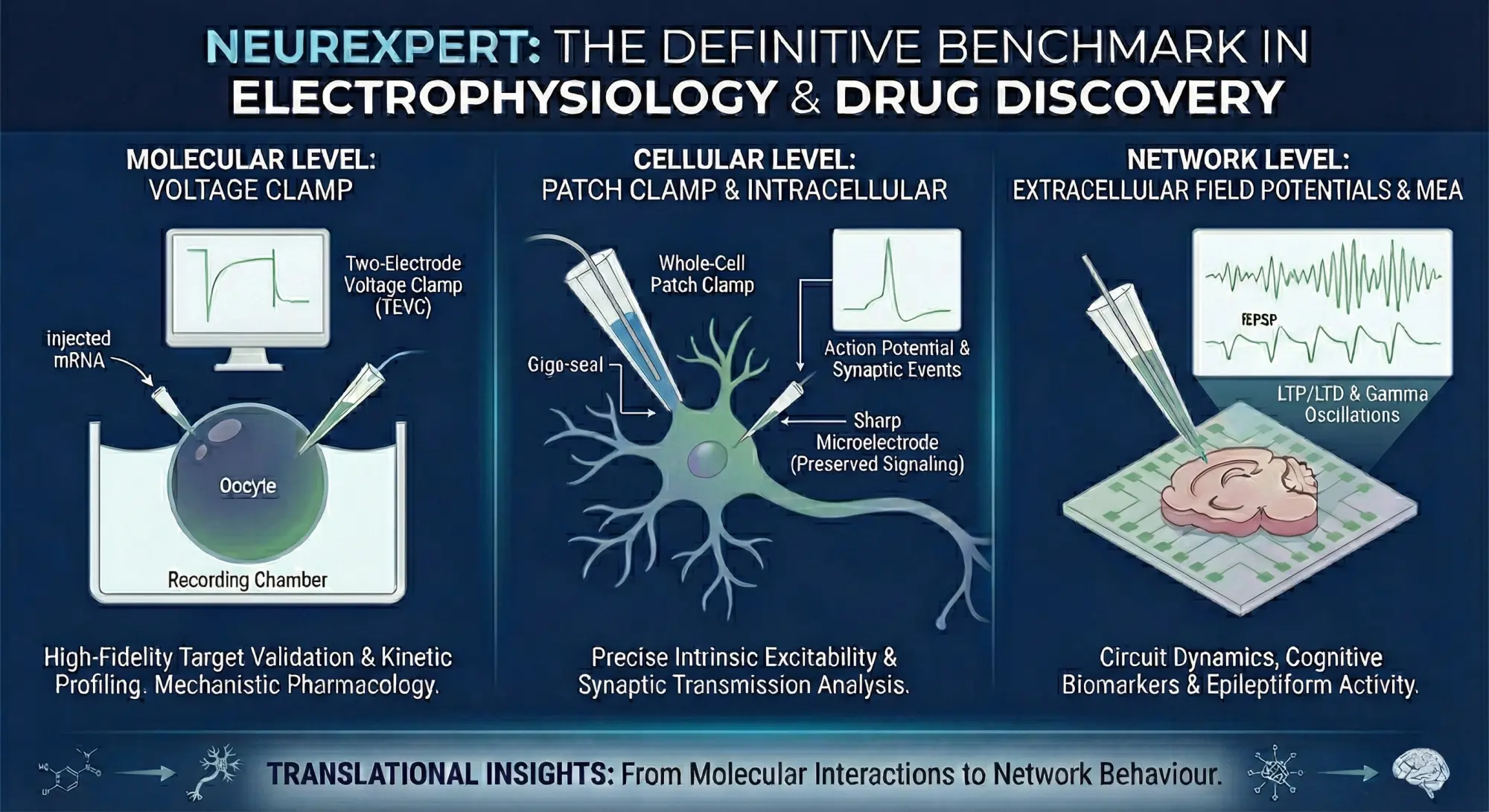
Molecular Level
Target Characterisation
Uncovering the precise pharmacological profile a target is the first step in rational drug design. We utilise high-fidelity electrophysiology to characterise ligand interactions, kinetics, and the functional consequences of genetic mutations with definitive precision.
Key Capabilities
- Functional Profiling of Diverse Targets: Comprehensive evaluation of ligand-gated and voltage-gated ion channels (LGICs/VGICs), G protein-coupled receptors (GPCRs), orphan receptors, enzyme modulators and electrogenic transporters.
- Advanced Pharmacological Characterisation: Precise quantification of compound potency (EC50/IC50), efficacy, and mechanistic modulation of receptor kinetics (activation, deactivation, and desensitisation), including the detailed characterisation of positive and negative allosteric modulators (PAMs/NAMs).
- Analysis of Complex Molecular Architectures: Investigating the functional impact of specific subunit stoichiometry, receptor co-expression, and the formation of receptor heterodimers.
- Optimisation of Challenging Targets: Expertise in expressing and recording from "difficult" proteins, including those with traditionally low expression levels or ultra-rapid desensitisation profiles.
- Cross-Species Comparison & Disease Modelling: Definitive comparison between human and rodent homologues, or functional profiling of rare patient-derived mutations (channelopathies) to support precision medicine.
- Secondary Screening & Hit-to-Lead Validation: Providing the critical functional insight required to de-risk assets before advancing them into more complex cellular and network-based models.
Technical Deep-Dive: Voltage Clamp recording & heterologous target expression
While automated high-throughput platforms are essential for initial library screening, the definitive characterisation of a lead compound requires the superior stability and precision of the Xenopus laevis oocyte system. It remains the definitive benchmark for mechanistic molecular pharmacology, providing the high-fidelity data required to transition from a "hit" to a "lead."
1. The "Biological Clean Room" for Target Validation
The Xenopus oocyte serves as a highly efficient heterologous expression factory. By injecting exogenous human or rodent mRNA/cDNA, we isolate your target in a noise-free environment.
- Absolute Target Specificity: We enzymatically or manually remove the follicular layer to eliminate endogenous interference. This ensures that every recorded picoampere is a direct readout of your compound's interaction with the specific target of interest.
- Precision Variant Modeling: This system is the premier tool for Precision Medicine and Channelopathy programmes. We can co-inject precise ratios of wild-type and mutant mRNA to model the exact heterozygous "gene-loading" found in patient populations.
2. The TEVC Advantage: Stability for Complex Pharmacology
The colossal size of the oocyte allows for Two-Electrode Voltage Clamp (TEVC), providing a level of recording stability that mammalian cells cannot match.
- Superior Dynamic Range: Expressed targets often generate massive currents (exceeding 10 μA). Neurexpert utilizes advanced bath-clamping and series resistance compensation to eliminate voltage-clamp errors, ensuring an accurate representation of compound efficacy even at high expression levels.
- Unrivalled Stability for Slow Kinetics: The longevity of oocyte recordings (often several hours) is vital for characterizing slow-binding, use-dependent, or irreversible inhibitors.
Cellular Level

Cellular Precision
Our patch-clamp and intracellular services assess the fundamental units of the nervous system. We move beyond simple recording to offer deep phenotypic and pharmacological profiling.
Key Capabilities
-
1. Neuronal Excitability & Intrinsic
Properties
We quantify the "firing fingerprint" of individual neurons to assess drug-induced changes in
intrinsic excitability and ion channel function.
- Passive Membrane Properties: Accurate determination of resting membrane potential (Vrest), input resistance (Rin), and membrane capacitance (Cm).
- Active Properties: Analysis of action potential (AP) threshold, rheobase (the minimum current required for an AP), and peak amplitude.
- Firing Kinetics: Characterisation of AP half-width, rise-time, and After-Hyperpolarisation (AHP) kinetics—critical for identifying modulators of Kv and SK channels.
- Repetitive Firing Patterns: Evaluation of frequency-current (f-I) relationships to identify compounds that normalize pathological firing (e.g., in epilepsy or pain models).
-
2. Synaptic Transmission &
Plasticity
We measure the efficacy and reliability of communication between neurons, identifying how
your lead compound "tunes" the synapse.
- Evoked & Spontaneous Activity: High-resolution analysis of EPSC/IPSC amplitudes, rise times, and decay kinetics.
- Quantal Analysis: Utilising Miniature Postsynaptic Currents (mEPSCs/mIPSCs) and Coefficient of Variation (CV) analysis to determine the biophysical properties of synaptic strength.
- Short-Term Plasticity (STP): Measurement of Paired-Pulse Ratios (PPR) and frequency-dependent facilitation or depression, revealing drug effects on neurotransmitter release probability (Pr).
- Tonic vs. Phasic Inhibition: Specialized assays for measuring the steady-state "tonic" inhibitory drive versus transient synaptic events.
-
3. Advanced Mechanism of Action
(MoA)
Neurexpert excels at de-risking compounds by pinpointing where and how they interact with
the neuronal architecture.
- Pre- vs. Post-Synaptic Locus: Definitive determination of whether a drug modulates neurotransmitter release (Presynaptic) or alters receptor sensitivity (Postsynaptic) using PPR and quantal metrics.
- Allosteric Modulation (PAMs/NAMs): Precise characterisation of Positive and Negative Allosteric Modulators, quantifying their ability to shift agonist potency and efficacy without blocking natural synaptic signaling.
- Second-Messenger Signaling: Leveraging sharp microelectrode recording to evaluate compounds targeting GPCRs (e.g., mGluR, Muscarinic, or GABA-B receptors) while keeping the intracellular signaling cascades (cAMP, IP3) intact.
Technical Deep-Dive: Patch Clamp & Intracellular
While high-throughput optical assays provide scale, electrophysiology remains the undisputed gold standard for understanding the precise pharmacology of ion channels, receptors, and transporters. At Neurexpert, we provide high-resolution, real-time measurements of protein activity within the complex architecture of native brain tissue.
1. Dual-Approach Intracellular Recording
Our laboratory utilises both patch-clamp (whole-cell) and sharp microelectrode techniques. This versatility allows us to select the optimal interface based on your drug target’s specific requirements.
- Whole-Cell Patch-Clamp: Provides unparalleled signal-to-noise ratios and precise control over the membrane potential. This is the primary tool for quantifying Miniature Postsynaptic Currents (mEPSCs/mIPSCs) and studying single-cell conductance changes. It also allows for the internal perfusion of drugs to target intracellular domains.
- Sharp Microelectrode Recording: A specialized technique essential for projects targeting G-Protein Coupled Receptors (GPCRs) and complex intracellular signaling (e.g., mGluR or Muscarinic pathways). Because the fine tip prevents the "washout" of the cytoplasm, we preserve the vital second-messenger systems (cAMP, IP3) that are often lost in standard patch-clamp configurations. This allows for stable, long-duration recordings required to assess slow-acting compounds.
2. Specialised Assays for Synaptic Function
We don't just record signals; we analyse the functional "logic" of the neuron:
- Dendritic Integration: Assessing how your compound modulates the way a neuron sums its inputs—a critical factor in cognitive enhancer development.
- Ionic Conductance Modulation: Precise characterisation of drug effects on specific currents such as Ih (hyperpolarisation-activated) or IK (potassium) conductances, which are key regulators of neuronal excitability and rhythmic activity.
Network Level

Network Dynamics
Bridging the gap to clinical efficacy. We investigate how compounds modulate complex network dynamics, oscillations, and enduring forms of synaptic plasticity (LTP/LTD) in native tissue, providing the most translationally relevant functional data available in vitro.
Key Capabilities
-
1. Synaptic Plasticity
(LTP/LTD)
We utilize extracellular field potential recordings to measure the cellular correlates of
learning and memory across distinct anatomical pathways.
- Hippocampal Circuitry: Extensive expertise in the Schaffer-collateral (CA3-CA1) pathway, as well as Mossy Fiber and Perforant Path inputs. We offer high-stability, long-term recordings to evaluate both the induction and maintenance phases of LTP.
- Cortical & Thalamocortical Pathways: Assessment of plasticity in the prefrontal and sensory cortex, providing a readout for drug candidates targeting executive function and sensory processing.
- Mechanism-Specific Induction: Selection of induction protocols (e.g., High-Frequency Stimulation, Theta-Burst, or Chemical LTP) tailored to the specific pharmacological profile of your compound.
-
2. Cognitive Biomarkers: Network
Oscillations
Higher-order cognitive functions emerge from synchronized neuronal firing. We quantify how
compounds modulate these critical rhythms.
- Induced Gamma-Frequency Activity: Utilizing pharmacological induction (e.g., Kainate or Carbachol), we evaluate the power, frequency, and synchrony of gamma oscillations (30–80 Hz), a key biomarker for information processing disrupted in Schizophrenia and Alzheimer’s disease.
- Theta-Gamma Coupling: Analysis of the interaction between slow (theta) and fast (gamma) rhythms, a sophisticated readout for cross-frequency coupling essential for spatial navigation and memory encoding.
- Spontaneous Oscillations: Measurement of endogenous rhythms, including Sharp-Wave Ripples (SWRs), to identify drug effects on the natural offline processing of information.
-
3. Circuit Stability & Epilepsy
Models
We offer a range of validated assays to test for both anti-epileptic efficacy and off-target
pro-convulsant liability.
- Ictal & Inter-ictal Discharges: High-resolution monitoring of spontaneous or induced paroxysmal activity. We characterize the frequency, duration, and spread of discharges across the network.
- Validated Seizure Models: Utilization of established in vitro models (e.g., 0-Mg2+, Picrotoxin, or 4-Aminopyridine) to simulate pathological hyperexcitability and evaluate the potency of novel Anti-Epileptic Drugs (AEDs).
- Phenotypic Rescue: Assessing the ability of lead compounds to normalize disrupted network activity in tissue derived from genetic models of channelopathies (e.g., Dravet Syndrome or KCNQ-related disorders).
Technical Deep-Dive: Extracellular Field Potentials
Extracellular field potential recordings are the definitive method for bridging the gap between molecular drug action and circuit-level behavior. By monitoring the synchronised activity of many neurones simultaneously, we provide a high-fidelity readout of synaptic efficacy, network stability, and emergent rhythms.
1. The Definitive Memory Assay: LTP and LTD
We specialize in quantifying Long-Term Potentiation (LTP) and Long-Term Depression (LTD)—the cellular foundations of learning and memory.
- Synaptic Plasticity: We measure the persistent change in fEPSP slope and amplitude following tetanic and high-frequency stimulation. This allows for the screening of "nootropic" compounds or the assessment of whether a drug candidate restores plasticity in disease models (e.g., Alzheimer’s or Fragile X).
- Baseline Efficacy (I/O): Before plasticity induction, we establish Input-Output (I/O) relationships—the ratio of the presynaptic fiber volley to the postsynaptic response. This baseline is critical for determining if a drug affects general synaptic transmission or specifically targets the mechanisms of plasticity.
2. Cognitive Biomarkers: Network Oscillations
Higher-order brain functions rely on specific electrical rhythms. We record and analyse these "biomarkers of cognition" in hippocampal and cortical slices:
- Gamma and Theta Oscillations: These rhythms are essential for information processing and attention. We test if drug candidates can normalize "dampened" oscillations associated with conditions such as Schizophrenia or ADHD.
- Sharp-Wave Ripples (SWRs): These highly synchronized events are the hallmark of memory consolidation during rest. Monitoring SWRs provides a unique window into how a drug might impact the "replay" and storage of information.
3. Safety Pharmacology & Epileptiform Analysis
Excitability is a delicate balance. Our field potential assays serve as a sensitive filter for pro-convulsant risks:
- Epileptiform Burst Analysis: By manipulating extracellular concentrations (e.g., 0-Mg2+ or high-K+), we induce seizure-like activity to evaluate the efficacy of novel anti-epileptic drugs (AEDs) or to screen for off-target hyperexcitability in new chemical entities.
- Complex Network Readouts: Unlike single-cell methods, field potentials capture the "recruitment" of neurons into pathological bursts, providing a more accurate prediction of in vivo seizure liability.
4. High-Density Spatial Mapping (MEA)
To increase throughput and spatial resolution, we utilise Multi-Electrode Arrays (MEA).
- Network Dynamics: Record from up to 96 sites simultaneously to map how a drug-induced signal propagates across different anatomical layers of a brain slice.
- Unbiased Screening: The array format allows for non-invasive, long-term monitoring of network health and drug-response stability over hours or even days.
Design Your Study
Select the level of analysis that fits your programme's stage. From target validation to IND-enabling network studies.
Consult with an Expert