Welcome to the Neurexpert Blog
Translating the Latest Neuroscience Research.
From biochemical pathways to modulation of brain oscillations
by Beth Dennis , 08 November 2017.
Tryptophan is one of the so-called essential amino acids that must be supplied in the diet of humans and other mammals. Tryptophan is needed for the synthesis of the neurotransmitter serotonin and the hormone melatonin, both of which are important in the regulation of mood, appetite, sleep and other essential brain functions. Tryptophan is also the precursor to N-formylkynurenine which, when converted by formamidase, produces kynurenine. This is the beginning of the kynurenine pathway, an enzymatic cascade responsible for over 90% of tryptophan metabolism, a source of NAD+ (a biologically necessary coenzyme) and a producer of a number of compounds which may play a role in various neurological and psychiatric diseases.
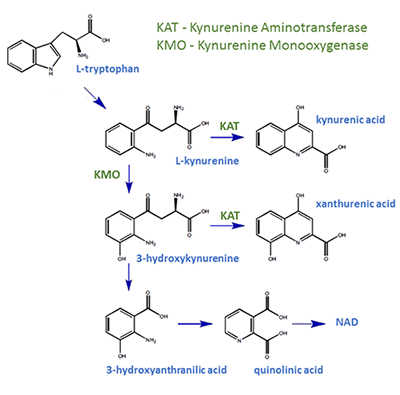
Advances in electrophysiological and biochemical techniques have put the kynurenine pathway and the products of this cascade in the spotlight. The kynurenine pathway produces many metabolites from tryptophan, including compounds such as kynurenic, quinolinic and xanthurenic acids, which are known to modulate the activity of neurones. Xanthurenic acid (XA), like quinolinic and kynurenic acids, is naturally occurring within the central nervous system, and results from the enzymatic reaction between kynurenine aminotransferases and, XA’s neurotoxic precursor, 3-hydroxykynurenine (3-HK). Recent research has characterised XA, identifying key features that could play a role in the pathophysiology of neurological and psychiatric disorders when the pathway malfunctions.
Increasingly, brain activity is being studied and manipulated to characterise and better understand the circuitry and mechanisms underlying cognitive impairments of brain related disease. Synchronous brain activity between 30 and 80 Hz, known as gamma oscillations, has been repeatedly associated with cognitive ability. Many studies, in vivo and in vitro, have shown altered gamma activity in schizophrenic patients and animal models of the disorder, as well as impairment in learning and memory.
Vesicular glutamate transporters (VGLUTs) are proteins that perform the uptake of glutamate, the principal excitatory neurotransmitter, into presynaptic vesicles. VGLUTs therefore play an important role in synaptic signalling. XA is an endogenous inhibitor of these VGLUTs, meaning the amount of glutamate being transported and released for neurotransmission is reduced when XA inhibits VGLUT. Attenuated VGLUT function, or expression, and reduced glutamate synaptic transmission are associated with deficits in sensory processing, learning and memory, all of which are symptoms associated with the cognitive impairments of schizophrenia.
In a recent study, Neurexpert collaborated with Dr. Korrapati Sathyasaikumar and Prof. Robert Schwarcz (Maryland Psychiatric Research Center) to evaluate the actions of XA and its precursor 3-HK. From this, Neurexpert demonstrated an effect on brain activity that is potentially relevant to known activity in the brain of schizophrenic patients. Using brain slice electrophysiology technology, bath application of the two kynurenine metabolites significantly reduced gamma oscillation power in the CA3 region of the hippocampus. This effect was seen to increase as the concentration of XA increased. In further experiments, 3-HK and XA were both found to reduce excitatory synaptic transmission in the hippocampus. The reduction in transmission caused by 3-HK decreased when the production of XA from 3-HK was inhibited by AOAA, confirming that increased XA reduces synaptic transmission. This evidence suggests that the xanthurenic acid arm of the kynurenine pathway plays an important role in the correct functioning of glutamatergic transmission and could participate in the pathophysiology of disorders such as schizophrenia. Examples of XA and 3-HK modulating gamma activity may be viewed in the presentation below.
But is there biological evidence for any of this found in patients? Yes; data shows that kynurenines, and particularly xanthurenic acid, levels are abnormal in schizophrenic patients compared to controls, and that kynurenine pathway metabolism is also dysfunctional in people with schizophrenia.
With all things considered, this evidence highlights xanthurenic acid as a functionally significant kynurenine pathway metabolite in the brain and, although there is still much to discover, it may offer a promising therapeutic target for the future. Furthermore, it is evident that recording gamma oscillations in vitro offers a useful translational functional assay for the development of drugs in the area of psychiatry.
Reference
Sathyasaikumar, K. V., M. et al. (2017). "Xanthurenic Acid Formation from 3-Hydroxykynurenine in the Mammalian Brain: Neurochemical Characterization and Physiological Effects." Neuroscience. doi:10.1016/j.neuroscience.2017.10.006
The Blog was written by Beth Dennis. To learn more about Beth and Neurexpert, please click here.
Recent Blog Posts
Astrocytes modulate thalamic sensory processing via mGlu2 receptor activation
Dancing with the stars: alternative target for Huntington’s disease treatment
Bioluminescence allows for optogenetics illumination of neurons with no excitatory light
Painful memories play role in chronic pain
Is chronic pain a problem with neural memory? We look at how the brain stores the pain and links it with psychological pain
Could photobiomodulation prevent the decline of bee populations caused by pesticides?
A look at a novel therapy for preventing drug seeking in addiction
One hurdle in overcoming addiction is ignoring cues in our environment that trigger drug seeking. We look at a new potential therapy for preventing drug seeking in addiction.
Targeting brain power to prevent and treat brain disorders
How disrupting brain energy processing can lead to brain disease- insights into how this can lead to a physiological fingerprint for diagnosis and drug development.
Neurexpert and Newcastle University to collaborate on translational drug discovery services
The power of touch and neurodevelopmental disorders
Touch is incredibly important for normal brain development- babies who are not touched have poor outcomes. But, what if a baby gets enough love but their skin nerve cells don’t process touch appropriately? Over 75% of patients with autism spectrum disorder suffer from a change in the way they perceive touch. We look at a Cell study that made a major breakthrough in understanding the connection between the skin and social and anxiety symptoms of neurodevelopment disorders.
Unsure if you are depressed? Could your gut makeup tell your doctor and help customize treatment?
Neurexpert collaborates with Cellectricon to provide enhanced CNS drug discovery opportunities
Neurexpert and Cellectricon will work together to promote combined CNS drug discovery

Brochure Publications